Research
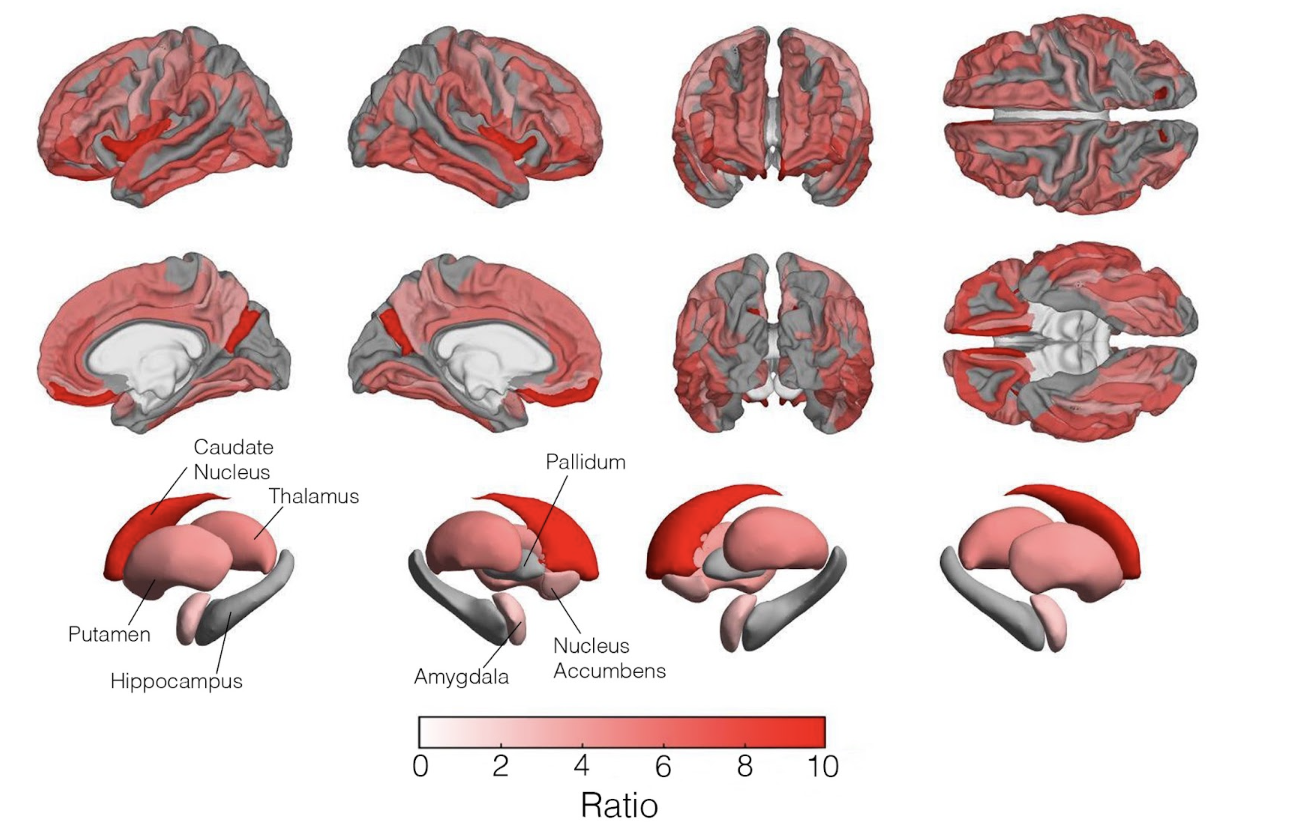
Brain age
In recent years, biological age has emerged as a useful metric for quantifying how an individual’s brain deviates from typical aging trajectories. The difference between an individual’s chronological age and predicted brain age, termed the brain age gap, is widely used as a marker of neurological injury, neurodegenerative disease, and disease risk. We develop and apply deep neural networks to estimate brain age from MRI data, considering both the overall pace of brain aging and regional variation in local brain age. These measures provide insight into atypical aging and have applications in assessing disease risk, including Alzheimer’s disease, as well as tracking cognitive recovery following traumatic brain injury.

Deep learning for neuroimaging and genetics
Artificial intelligence is one of the fastest growing fields in research and continues to expand rapidly across disciplines. Our lab develops explainable AI methods to analyze neuroimaging and genomic data, with the goal of advancing our understanding of healthy aging and neurodegenerative disease. Our models leverage cutting edge deep learning and multimodal data integration techniques to capture complex relationships between brain structure, genetics, cognition, and aging. Through these efforts, we aim to bridge computational innovation with clinical and neuroscientific applications, contributing both methodological advances and a deeper understanding of brain health across the lifespan.
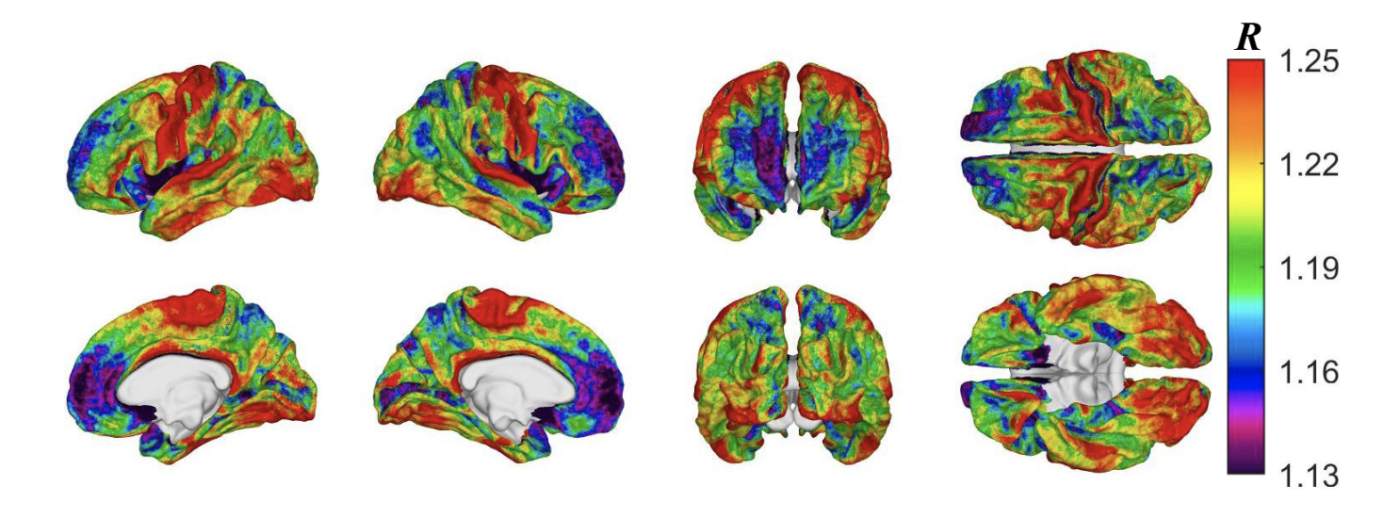
Demyelination in health and disease
Myelin sheaths are fatty axonal coatings that are essential to the health and function of the brain and play a key role in both normal aging and disease. Cortical demyelination can occur in traumatic brain injury (TBI) and is also associated with broader neurodegenerative processes. The ratio R of T1– to T2-weighted magnetic resonance image intensities serves as a measure of relative myelin content in the cortex, which we can compute longitudinally across clinical cohorts. Using this approach, we map intracortical myelin and its changes over time, comparing patterns of demyelination after mTBI to those seen in typical aging to better understand central nervous system dysfunction and inform potential remyelination therapies.

Functional neuroanatomy of brain injury
Studying the functional neuroanatomy of mild traumatic brain injury (mTBI) is essential for understanding neurological and cognitive outcomes post-injury. The brain undergoes significant changes in functional connectivity (FC), quantified using functional magnetic resonance imaging (fMRI), after mTBI, and these alterations underlie persistent neural and cognitive effects that vary with age and sex. Our work focuses on post-traumatic FC changes across both acute and chronic stages of injury, mapping sex- and age-dependent patterns across cortical resting-state networks (RSNs) in a cohort of mTBI patients. Our findings indicate that male sex and older age at injury are associated with more pronounced FC alterations, which may contribute to post-traumatic cognitive deficits.
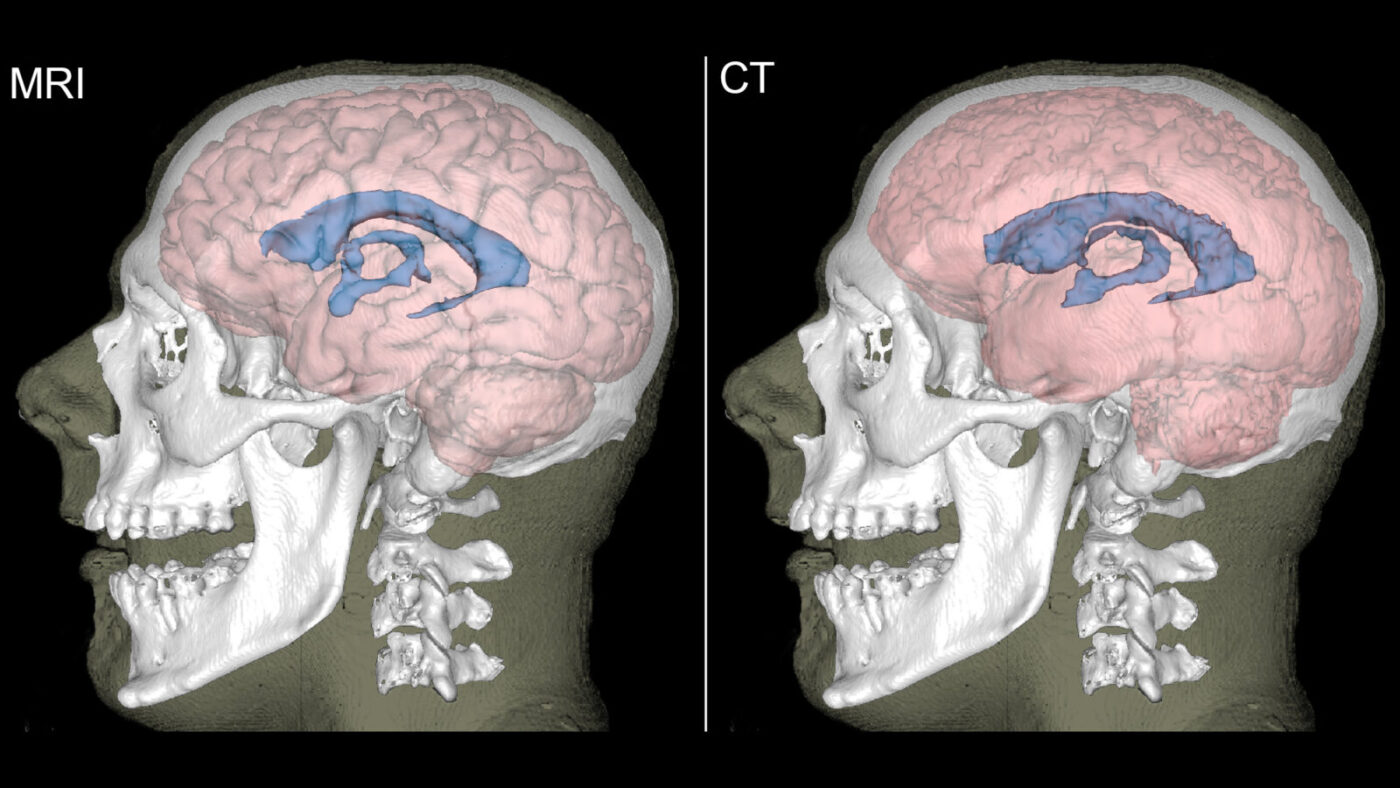
Atypical aging in non-industrial populations
Accelerated brain loss and atrophy can indicate increased risk of dementia. The indigenous forager-horticulturist Tsimané of lowland Bolivia show a decline in brain volume that is about 70% slower than that of Western populations. We generate automated segmentations of computed tomography (CT) brain scans from the Tsimané Health and Life History Project, enabling volumetric analysis of subcortical and cortical regions to compare brain development and atrophy with populations in developed countries. Because of their non-sedentary lifestyle and diet rich in fiber, vegetables, and lean meats, the Tsimané provide a unique opportunity to study the effects of modern lifestyles on brain health and aging.
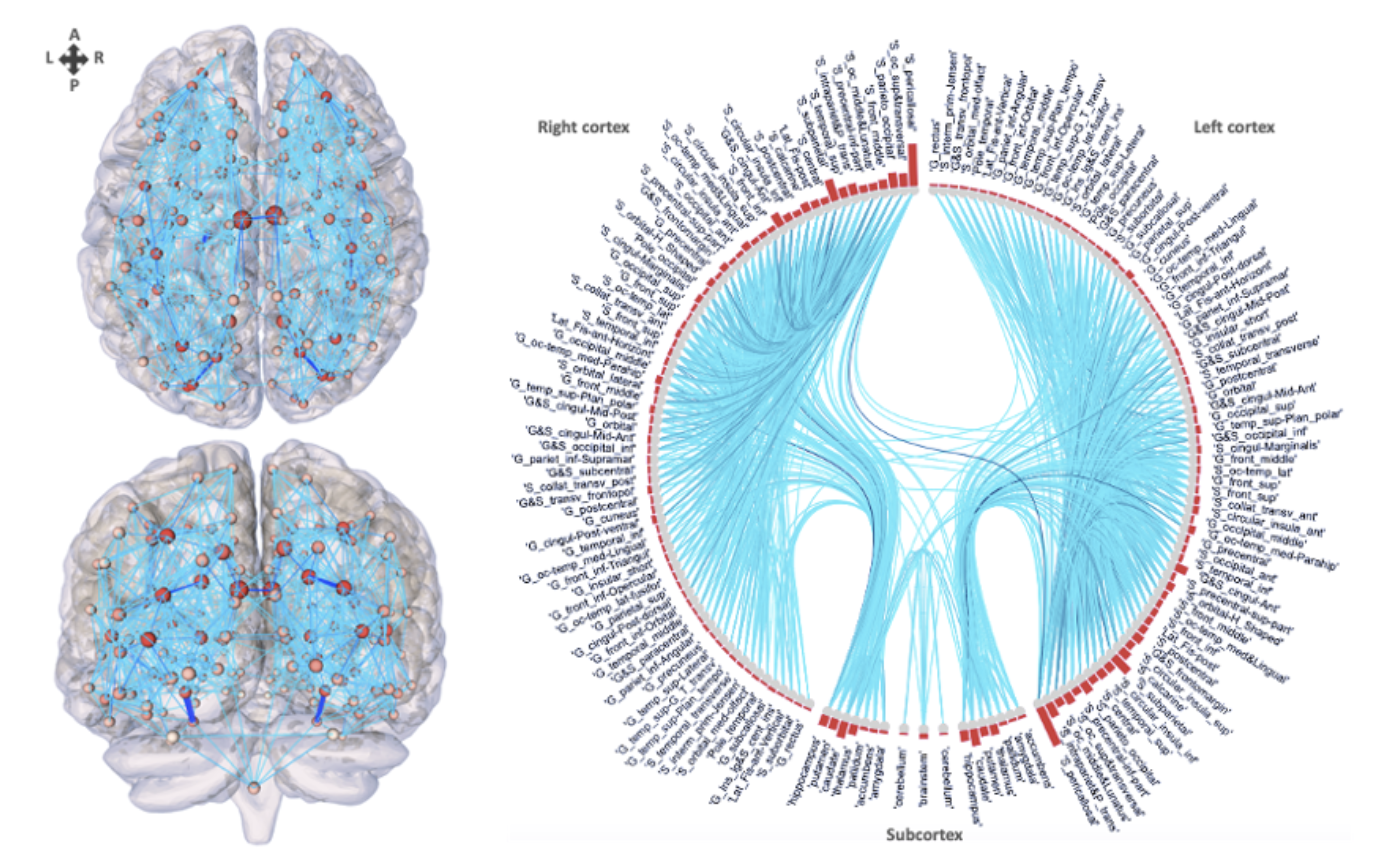
Structural connectomics in health and disease
Mild traumatic brain injury (mTBI) can lead to a range of cognitive and neurological effects that often persist despite normal findings on conventional magnetic resonance imaging (MRI). Damage to white matter, caused by diffuse axonal injury or secondary chemical processes such as excitotoxicity, may not be visible on these scans. Our team uses diffusion MRI (dMRI) and network theory to characterize brain networks in individuals with mTBI, exploring structural connectomics as an indicator of cognitive outcomes by extracting quantitative measures of brain connectivity. We aim to track changes in network organization and cognitive function over time in individuals with mTBI compared to healthy controls, using machine learning and statistical methods to evaluate the predictive value of these network metrics for recovery and decline.

