Research
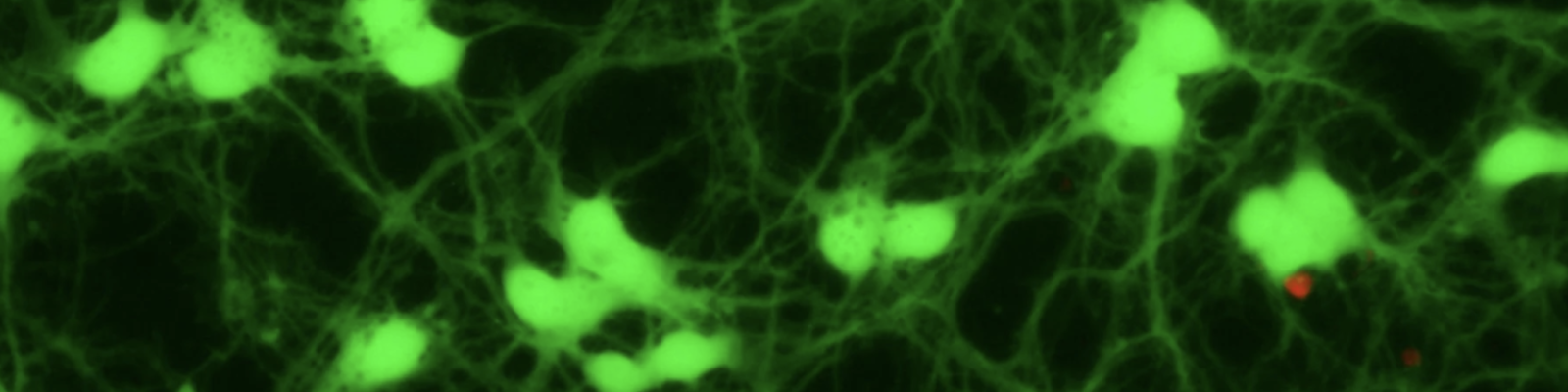
Ongoing research projects in the Pike Lab are using a range of rodent models, cell culture paradigms and analyses of human tissues both to investigate underlying factors in Alzheimer’s pathogenesis and to develop translatable approaches to prevent and treat the disease. Current investigations, outlined below, emphasize the interactive risks of obesity and depletion of sex steroid hormones as well as pursue two promising, novel therapeutic strategies.
Sex differences in AD risk associated with APOE4
APOE4, the primary genetic risk factor for Alzheimer’s disease (AD), causes a greater lifetime risk and an increased rate of cognitive decline in women compared to men. The reasons for this sex-based link remain unknown. Our collaborative team is investigating the novel hypothesis that normal developmental processes that underlie sexual differentiation yield a female brain that is inherently more vulnerable to the effects of APOE4. We are examining this hypothesis using both human twin populations and transgenic mice that harbor human APOE alleles and AD transgenes. The results of this research will provide essential information to address the critical need for treatment options for women at high risk for AD from APOE4.
Protective diets as prevention and treatment strategies against APOE4 and Alzheimer’s disease
The E4 allele of apolipoprotein E (APOE4) is associated with accelerated aging and mortality as well increased vulnerability to Alzheimer’s disease (AD). Although the causal links between APOE4, aging, and AD risk remain to be fully defined, candidate mechanisms include regulation of energy metabolism and systemic and neural inflammatory tone. Recent findings from our collaborator Valter Longo demonstrate that nutritional approaches, including fasting mimicking diet (FMD), exhibit anti-aging and protective properties across several age-related conditions. Indeed, pathways identified to be beneficially regulated by FMD and related nutritional interventions overlap with pathways thought to underlie relationships among APOE4, aging, and AD. In an ongoing series of studies, we investigate the hypothesis that FMD and related dietary interventions will protect against APOE4 phenotypes that drive age-related cognitive impairment and AD pathogenesis.
Longevity-promoting compounds as protectants against APOE4 and Alzheimer’s disease
Increasing age is the most significant risk factor for developing Alzheimer’s disease (AD). APOE is the most common genetic AD risk factor: AD risk is reduced by the APOE2 allele and increased by the APOE4 allele. Interestingly, APOE genotype is associated with longevity, whereby APOE2 is linked with increased and APOE4 decreased longevity. This same pattern is observed with AD, in which risk follows an APOE2 < APOE3 < APOE4 pattern. An intriguing possibility is that the relationships between AD risk and APOE genotypes are mediated in part by regulation of systemic aging. Thus, age-related phenotypes associated with APOE4 are predicted to contribute to its increased AD risk. Indeed, aging and APOE4 share several phenotypes that are linked with AD pathogenesis, including systemic and neural metabolic dysfunction and inflammation. We are actively pursuing several studies investigating the hypothesis that the longevity-promoting interventions will preferentially protect against APOE4 phenotypes that drive AD pathogenesis. These studies have potentially profound impact in that they suggest the possibility that senescent, AD-related phenotypes associated with APOE4 may be prevented by longevity-promoting treatments in middle age.

